 The EU Falsified Medicines Directive will introduce major changes for community pharmacists across the UK when its requirements for medicines verification are introduced from 9th February 2019. This page sets out the background to FMD.
The EU Falsified Medicines Directive will introduce major changes for community pharmacists across the UK when its requirements for medicines verification are introduced from 9th February 2019. This page sets out the background to FMD.
The European Falsified Medicines Directive [2011/62/EU] (commonly known as FMD) has been introduced to address the issue of falsified medicines entering the legitimate medicines supply chain across Europe. Falsified medicines include those with little or no active ingredients, the wrong active ingredients, fake or tampered packaging, and those where products and/or packaging have been stolen for re-use or re-sale.
Although unlicensed internet sites have now become the main route for trading falsified medicines to the public, regulatory authorities continue to find and report falsified medicines in the supply chain. Such products pose an unacceptable risk to patients, as well as undermining confidence in the genuine products supplied by manufacturers, wholesalers and pharmacies.
Under the Directive, all new packs of prescription medicines placed on the market in Europe from 9th February 2019 onwards will have to bear two safety features: a unique identifier (UI) in the form of a 2D data matrix (barcode) and an anti-tampering device (ATD).
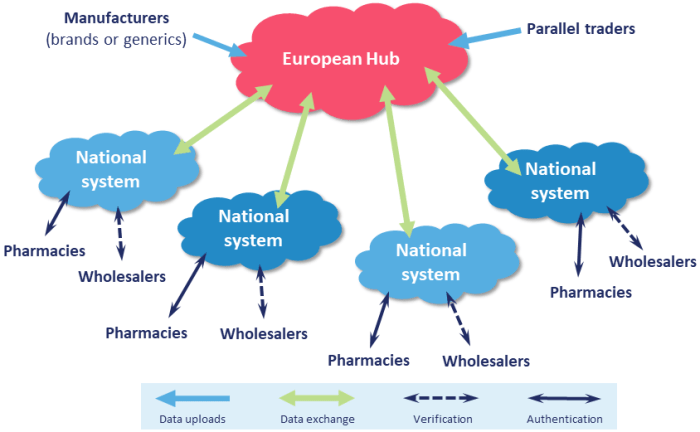
FMD will introduce a pan-European system through which pharmacies (and other “dispensing entities”) can authenticate prescription medicines before they are handed to patients. Manufacturers will upload valid UI codes to a European Hub, which will then pass data to the relevant National Medicines Verification System (NMVS) and pharmacies will be able to check the status of each pack during the dispensing process. The system will be notified of products known to have been recalled, withdrawn, stolen or tampered with. The European Medicines Verification System (EMVS) will act as the European hub, linking the national systems together and allowing parallel trading of medicines to continue.
Implementing FMD
Scanning and authentication of packs will come in to effect across Europe from Saturday 9th February 2019, following the publication of a Delegated Regulation [2016/161] which set out details for the unique identifiers, the national verification systems and the responsibilities of manufacturers, wholesalers and pharmacies.
FMD will be introduced in 32 countries – the 28 (current) Member States of the EU, plus the four members of the European Free Trade Area (Iceland, Norway, Liechtenstein and Switzerland).
Policy work on FMD has been going on for around a decade, leading up to the adoption of the EU Directive in 2011. Pharmacy bodies, including the National Pharmacy Association and the Royal Pharmaceutical Society were involved in this through the Pharmaceutical Group of the European Union (PGEU), the European Union pharmacy association. Further work on technical details of serialisation and the verification systems saw the Delegated Regulation published in February 2016. It will come into force across Europe from 9th February 2019.
The development of the European and national verification systems is being led through a stakeholder model involving the five main sectors (or “constituencies”) of the supply chain – research-based manufacturers, generic manufacturers, parallel traders, wholesalers and pharmacies – working alongside the European Commission and national competent authorities (Health Ministries and regulators).
At a European level, the European Medicines Verification Organisation (EMVO) has been established by the European associations representing the five sectors. The EMVO has developed a set of governing principles that all parties have agreed to. Some of the key principles relate to data security, including:
- Data ownership is determined by the party who generates the data;
- Data in the system is not to be used to analyse the flow of products in the supply chain;
- The system allows for verification of products at any point but is not a “track-and-trace” system and does not give visibility of current or past locations of products; and
- Manufacturers will not seek, and will not have access to, individual prescribing or patient profile information.
As well as establishing the central European Medicines Verification System (EMVS), or European hub, which is now up and running, the EMVO has developed the blueprint model for national verification systems. This is designed to promote harmonisation and reduce costs.
Under the blueprint model, three companies were chosen to bid for Blueprint Service Provider (BSP) contracts to run national medicines verification systems. These were Aegate, Arvato and Solidsoft Reply. Each has experience in providing large-scale IT projects, including serialisation. Since the start of the process, Aegate has withdrawn from the market.
National stakeholder associations across Europe have worked to establish their own National Medicines Verification Organisations (NMVO); the UK NMVO is SecurMed UK, which comprises bodies representing manufacturers, importers, wholesalers and pharmacies. Community pharmacy is represented on SecurMed UK jointly by the National Pharmacy Association and the Company Chemists’ Association.
SecurMed UK selected Arvato as their Blueprint Service Provider and they entered into a contract for provision of the UK FMD system in July 2018.
The scope of FMD
Almost all prescription medicines with a Marketing Authorisation are within the scope of FMD, with only a few specialist products, radionuclide kits and medicinal gases being out-of-scope. All non-prescription medicines are out-of-scope, with the exception of two omeprazole products (which were subject to falsification in the past).
Unlicensed products, including specials and clinical trial supplies, are out-of-scope, but any licensed products being incorporated into them (such as ingredients for specials) would have to be authenticated and decommissioned before they could be used.
Medical devices are out-of-scope, but separate changes are being made to regulation of medical devices.
Unique identifiers
Each individual pack of a prescription medicine will need to carry a unique identifier (UI) encoded via a 2D data matrix (barcode). If the pack size permits it, the pack will also carry the same information in human-readable text, printed adjacent to the 2D-code.
The unique identifier will consist of:
- Product code: the name, common name, pharmaceutical form, strength, pack size and pack type
- Serial number: randomised numeric or alphanumeric sequence of up to 20 characters
- National reimbursement number: national identifying code, if required by the Member State
- Batch number: up to 20 characters
- Expiry date: in YYMMDD form
Because this information will be printed and encoded on every pack, it can also be used for some activities in pharmacy, such as stock re-ordering, stock rotation and accuracy checking.
How FMD will work
Each manufacturer will generate randomised unique identifiers and print these on relevant packs of medicines it produces, as a 2D data matrix and (space permitting) in human readable form. All the unique identifiers for a batch of product will be uploaded by the manufacturer to the European Hub when the product is released for sale. The UIs will then have an “active” status.
The European Hub will validate the UI data and transfer it to the relevant National Medicines Verification System(s). Pharmaceutical wholesalers, community and hospital pharmacies, dispensing medical practices and any other persons “authorised to supply medicines to the public” will connect to their local NMVS.
As part of the dispensing process, pharmacies will scan the UI and send the details to the NMVS, which will compare the UI with the data uploaded by manufacturers; this process is known as verification. If the two items match, a confirmation message will be returned. The dispenser will then send another message to the NMVS to change the status of the product to “Inactive – decommissioned”, preventing any other pack with the same UI from being authenticated – duplication of packs being a sign that falsification might have occurred. This process is known as decommissioning. During dispensing, there will also be a check that the pack still has an intact anti-tampering device.
The FMD system can also be set to stop the dispensing of products that have been withdrawn, recalled or which are known to be stolen.
Parallel traders will also be able to use the system. They will decommission the UIs of products to be repacked or relabelled and then upload new UIs (maintaining a link at batch level to the original product).
